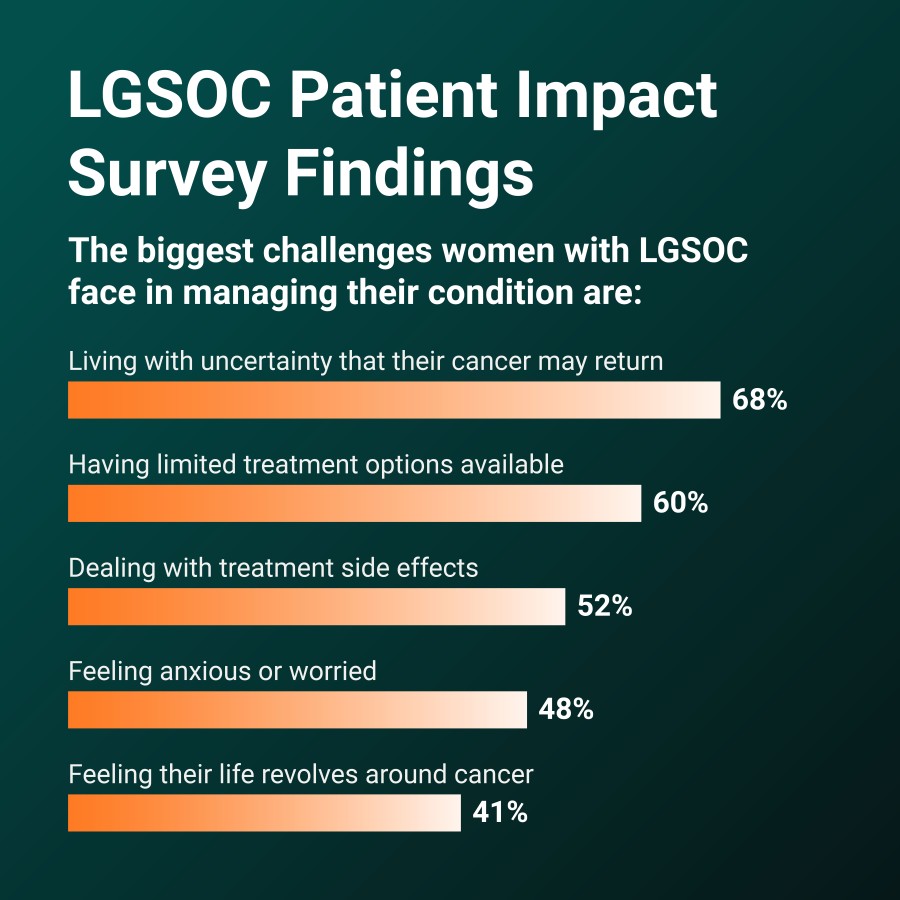
John Johnson, current board member, appointed to chairman of the board Michael Kauffman, M.D., Ph.D., lead director since 2016, appointed to president of development Commercial launch progresses as RAMP 301 Phase 3 confirmatory trial in recurrent LGSOC completes additional patient enrollment; topline data anticipated in mid-2027 BOSTON / Dec 15, 2025 / Business Wire / Verastem Oncology (Nasdaq: VSTM), a biopharmaceutical... Read More